Health
Eli Lilly Secures IL-6 Antibody from CSL for $100 Million Upfront

Eli Lilly has made a significant move in the pharmaceutical sector by acquiring CSL Limited’s IL-6 immuno-inflammatory antibody for an upfront payment of $100 million. This deal marks an important step for both companies, particularly as CSL navigates ongoing financial challenges.
The IL-6 antibody, which targets interleukin-6, is seen as a promising asset in the treatment of various inflammatory conditions. By securing this licensing agreement, Eli Lilly aims to enhance its portfolio in immunology, demonstrating a strategic focus on innovative therapies that address unmet medical needs.
CSL Limited, an Australian biotechnology company, is currently facing difficulties that have prompted this licensing agreement. The financial injection from Eli Lilly will assist CSL in stabilizing its operations while continuing to develop its extensive pipeline of therapies.
Strategic Implications of the Deal
This licensing agreement not only provides immediate financial relief for CSL but also sets the stage for future collaboration between the two companies. Eli Lilly’s expertise in drug development and market access could accelerate the IL-6 antibody’s pathway to patients.
CSL has previously seen significant successes with its treatments for serious medical conditions, and this partnership could leverage their combined strengths. The financial commitment from Eli Lilly underscores the confidence in the potential of the IL-6 antibody to provide beneficial outcomes for patients suffering from chronic inflammatory diseases.
For Eli Lilly, this acquisition represents a vital addition to its growing portfolio, which has been bolstered by recent advancements in immunotherapy. The company is keen to expand its reach in immunology, and the IL-6 antibody aligns well with its strategic goals.
Market Reactions and Future Outlook
Market analysts have responded positively to the news, highlighting the potential for both companies to gain traction in the competitive biopharmaceutical landscape. The deal, finalized in early October 2023, is expected to enhance Eli Lilly’s market position while offering CSL the necessary resources to navigate its current challenges.
As both companies move forward with this agreement, the healthcare community will be watching closely to see how the IL-6 antibody progresses through clinical development. The collaboration could pave the way for innovative treatments that address significant health issues, benefiting patients worldwide.
Overall, the licensing of CSL’s IL-6 antibody to Eli Lilly not only reflects a strategic business decision but also underscores the dynamic nature of the pharmaceutical industry, where partnerships can lead to groundbreaking advancements in healthcare.
-

 Lifestyle4 months ago
Lifestyle4 months agoSend Holiday Parcels for £1.99 with New Comparison Service
-

 Science5 months ago
Science5 months agoUniversity of Hawaiʻi Leads $25M AI Project to Monitor Natural Disasters
-

 Top Stories4 months ago
Top Stories4 months agoMaui County Reopens Upgraded Lānaʻi Fifth Street Courts Today!
-
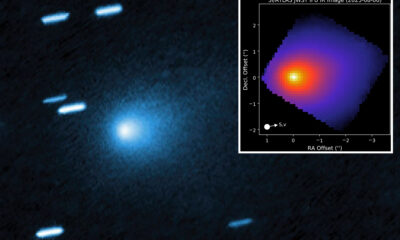
 Science6 months ago
Science6 months agoInterstellar Object 3I/ATLAS Emits Unique Metal Alloy, Says Scientist
-

 Entertainment6 months ago
Entertainment6 months agoKelly McCreary Discusses Future of Maggie and Winston in Grey’s Anatomy
-

 Entertainment6 months ago
Entertainment6 months agoDaily Codeword Puzzle Launches on October 21, 2025
-

 Lifestyle5 months ago
Lifestyle5 months agoCongresswoman Under Fire for Misleading Epstein Donation Claims
-

 Top Stories5 months ago
Top Stories5 months agoTrump Vows to Resolve Afghanistan-Pakistan Crisis “Very Quickly”
-

 Science6 months ago
Science6 months agoResearchers Achieve Fastest Genome Sequencing in Under Four Hours
-

 Business6 months ago
Business6 months agoIconic Sand Dollar Social Club Listed for $3 Million in Folly Beach
-

 Science4 months ago
Science4 months agoCharles Darwin’s Address Book Reveals Hidden Aspects of His Life
-

 Politics6 months ago
Politics6 months agoAfghan Refugee Detained by ICE After Asylum Hearing in New York