Health
CapsoVision Examines Position Against Competitors in Medical Sector

CapsoVision, a medical technology company listed on NASDAQ under the ticker symbol CV, is assessing its competitive standing within the “Surgical, Medical, and Dental Instruments and Supplies” industry. This analysis compares CapsoVision’s performance metrics, including profitability, analyst ratings, valuation, and institutional ownership, against its peers.
Profitability and Valuation Insights
CapsoVision’s financial indicators reveal a varied landscape when compared to its competitors. The company’s net margins, return on equity, and return on assets are critical metrics that highlight its profitability. While specific figures are not disclosed in this overview, they play a significant role in investor evaluations.
In terms of valuation, analysts have set a consensus price target of $6.00 for CapsoVision, indicating a potential upside of 18.58%. This contrasts sharply with the broader sector, where companies in the same category have a collective upside potential of 56.47%. Analysts suggest that this disparity may reflect a less favorable growth outlook for CapsoVision compared to its industry peers.
Market Analysis and Ownership Trends
Institutional investors hold approximately 19.3% of shares across the “Surgical, Medical, and Dental Instruments and Supplies” sector, while company insiders own 19.5%. Strong institutional ownership often signals confidence from large investors, indicating a belief that the stock will outperform the market over time.
In summary, CapsoVision has underperformed against its competitors in six of the eleven metrics examined, raising questions about its market positioning and growth prospects relative to others in the industry.
CapsoVision specializes in advanced imaging and artificial intelligence technologies for capsule endoscopy, primarily targeting the gastrointestinal (GI) tract. The company’s flagship product, the CapsoCam Plus, is a wire-free capsule designed to visualize the small bowel and identify abnormalities such as obscure GI bleeding and Crohn’s disease.
The device captures video images as it navigates through the GI tract, with the data stored in onboard memory. Healthcare providers can access these images via the CapsoCloud software or the CapsoView application, enhancing convenience and efficiency in diagnosis.
The CapsoCam Plus is classified as a Class II medical device and received FDA marketing authorization through the 510(k) clearance process. CapsoVision is currently working on enhancing this system with AI-assisted reading technology, aiming for FDA and EU submissions in the latter half of 2025.
The AI tools are designed to assist clinicians by highlighting potential abnormalities, thereby reducing the time needed for video analysis and making the procedure more appealing economically. Nonetheless, the company faces risks, as delays in the FDA review process could hinder timely approvals and market entry.
CapsoVision began international sales in 2012 and entered the U.S. market in 2017. Its sales strategy includes direct sales in the U.S. and partnerships with exclusive distributors internationally, with key markets identified as France, Germany, and Canada. The establishment of a direct sales team in Germany in 2023 aims to bolster its market presence in this significant region.
Revenue growth has been a positive trend for CapsoVision, with reported revenues of approximately $9.8 million for the year ending December 31, 2023, expected to rise to about $11.8 million in 2024, marking a year-over-year increase of 21%. For the first quarter of 2024 and 2025, revenues reached approximately $2.5 million and $2.8 million, respectively, an increase of 12% year-over-year.
The growth is primarily driven by a 19% increase in CapsoCam Plus capsule sales from 2023 to 2024, with unit sales rising 26% in the U.S. and 4% internationally. International sales contributed 26% and 23% to total revenue in 2023 and 2024, respectively, indicating a steady international customer base.
Looking forward, CapsoVision aims to expand its product line with the CapsoCam Colon, designed for colon examinations. This new capsule will incorporate AI features to detect polyps, a critical advancement as polyp size is directly linked to cancer risk. The company targets revenue generation from the CapsoCam Colon in the U.S. by the second half of 2026 following FDA clearance.
Despite the promising developments, CapsoVision’s growth strategy hinges on its ability to meet cash flow needs and navigate regulatory approvals effectively. Given its history of operating losses and an accumulated deficit, the company’s future remains closely tied to its financial health and market execution strategies.
CapsoVision, incorporated in Delaware on August 1, 2005, has undergone significant evolution, including a name change to CapsoVision, Inc. on May 31, 2016. With its principal office located in Saratoga, California, the company is poised to continue its efforts in advancing GI diagnostic technologies while seeking to improve its competitive standing in the medical device industry.
-

 Lifestyle4 months ago
Lifestyle4 months agoSend Holiday Parcels for £1.99 with New Comparison Service
-

 Science5 months ago
Science5 months agoUniversity of Hawaiʻi Leads $25M AI Project to Monitor Natural Disasters
-

 Top Stories4 months ago
Top Stories4 months agoMaui County Reopens Upgraded Lānaʻi Fifth Street Courts Today!
-
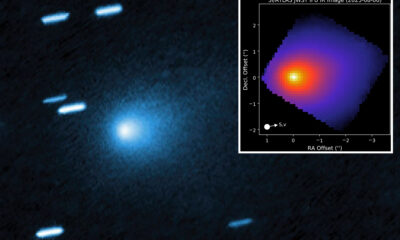
 Science6 months ago
Science6 months agoInterstellar Object 3I/ATLAS Emits Unique Metal Alloy, Says Scientist
-

 Entertainment6 months ago
Entertainment6 months agoKelly McCreary Discusses Future of Maggie and Winston in Grey’s Anatomy
-

 Entertainment6 months ago
Entertainment6 months agoDaily Codeword Puzzle Launches on October 21, 2025
-

 Lifestyle5 months ago
Lifestyle5 months agoCongresswoman Under Fire for Misleading Epstein Donation Claims
-

 Top Stories5 months ago
Top Stories5 months agoTrump Vows to Resolve Afghanistan-Pakistan Crisis “Very Quickly”
-

 Science6 months ago
Science6 months agoResearchers Achieve Fastest Genome Sequencing in Under Four Hours
-

 Business6 months ago
Business6 months agoIconic Sand Dollar Social Club Listed for $3 Million in Folly Beach
-

 Politics6 months ago
Politics6 months agoAfghan Refugee Detained by ICE After Asylum Hearing in New York
-

 Science4 months ago
Science4 months agoCharles Darwin’s Address Book Reveals Hidden Aspects of His Life